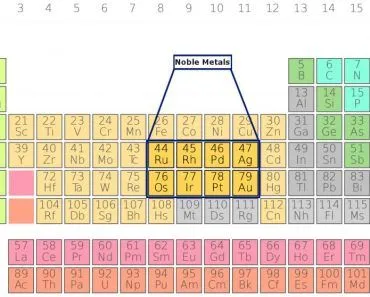
Aqua regia is a highly corrosive acid mixture formed by combining concentrated nitric acid (HNO₃) and concentrated hydrochloric acid (HCl) in a molar ratio of 1:3. The term ‘aqua regia’ is Latin for ‘royal water’ — a name given by medieval alchemists because this mixture could dissolve the ‘noble’ metals, such as gold and platinum, which resist attack by individual acids.
If you’re a chemistry enthusiast, you probably know that gold is an incredibly inert metal. It doesn’t rust, tarnish, or react with most chemicals. In fact, this chemical stubbornness is a big part of why gold has been so highly valued throughout history — it literally lasts forever. So, what could possibly dissolve something so resistant?
The answer is aqua regia — a fuming, corrosive liquid that has fascinated chemists and alchemists for over a thousand years. The earliest known description of aqua regia is attributed to the works of the Arab-Persian alchemist Jabir ibn Hayyan (also known as Geber) around the 8th century. European alchemists encountered it through Latin translations of Arabic texts and were captivated by its ability to dissolve the “king of metals.”
Aqua Regia Formula And Recipe
Aqua regia is prepared by mixing concentrated nitric acid and concentrated hydrochloric acid in a molar ratio of 1:3 (one part HNO₃ to three parts HCl). When freshly prepared, aqua regia is colorless, but it rapidly turns yellow, orange, or red within seconds due to the formation of reactive chemical species — specifically nitrosyl chloride (NOCl) and chlorine gas (Cl₂).
The decomposition reaction can be written as:
HNO₃ (aq) + 3 HCl (aq) → NOCl (g) + Cl₂ (g) + 2 H₂O (l)
These freshly generated reactive species — nitrosyl chloride and nascent chlorine — are what give aqua regia its extraordinary dissolving power. The mixture should always be prepared fresh before each use, as it rapidly loses its effectiveness. When preparing aqua regia, the nitric acid must always be added to the hydrochloric acid slowly, and the mixture should only be handled in a fume hood due to the release of toxic and corrosive gases.
How Does Aqua Regia Dissolve Gold?
Neither nitric acid nor hydrochloric acid can dissolve gold on its own. Nitric acid is a powerful oxidizer, but gold is too electrochemically noble to be oxidized by it alone. Hydrochloric acid provides abundant chloride ions, but has no oxidizing power to ionize gold. However, when the two are combined, a remarkable synergy occurs that makes the dissolution possible.
The process works in two coordinated steps:
Step 1 — Oxidation: Nitric acid acts as a powerful oxidizing agent, converting a small amount of metallic gold (Au) into gold ions (Au³⁺). On its own, this reaction barely proceeds because the equilibrium strongly favors the metallic form.
Step 2 — Complexation: This is where hydrochloric acid plays its crucial role. The chloride ions (Cl⁻) from HCl immediately react with the Au³⁺ ions to form tetrachloroaurate(III) anions, [AuCl₄]⁻. This stable complex effectively removes free Au³⁺ from the solution, shifting the equilibrium and allowing more gold to be oxidized.
The overall balanced equation for the dissolution of gold is:
Au (s) + 3 HNO₃ (aq) + 4 HCl (aq) → HAuCl₄ (aq) + 3 NO₂ (g) + 3 H₂O (l)
The product, chloroauric acid (HAuCl₄), is a soluble gold compound that gives the solution its characteristic deep yellow-orange color. This is essentially how gold “disappears” into the liquid — it hasn’t been destroyed, but rather converted into a dissolved ionic form.
Aqua Regia And Platinum
Platinum, another famously inert noble metal, is also dissolved by aqua regia through a similar mechanism. The reaction produces chloroplatinic acid (H₂PtCl₆):
3 Pt (s) + 4 HNO₃ (aq) + 18 HCl (aq) → 3 H₂PtCl₆ (aq) + 4 NO (g) + 8 H₂O (l)
However, it is important to note that aqua regia does not dissolve all noble metals. Metals such as iridium, osmium, tantalum, and titanium resist its attack. Silver is also not dissolved — instead, it forms a layer of insoluble silver chloride (AgCl) on its surface, which actually protects it from further reaction.
Aqua Regia Uses And Applications
Aqua regia has several important practical applications:
- Gold refining: In the Wohlwill process, aqua regia is used to create a solution of chloroauric acid, which serves as the electrolyte for producing gold of very high purity (99.99%).
- Microelectronics and microfabrication: Aqua regia is widely used to remove noble metals such as gold and platinum from substrates, making it an essential tool in semiconductor and microelectronics labs.
- Laboratory glassware cleaning: It is used to remove trace metal residues from labware, particularly NMR tubes and other sensitive analytical equipment.
- Analytical chemistry: Aqua regia is used to dissolve metal samples for quantitative chemical analysis, especially in environmental testing and geological surveying.
- E-waste recycling: Aqua regia plays a role in recovering precious metals from electronic waste, including circuit boards and electronic components.
Aqua Regia And Its World War 2 Story
Perhaps the most famous story involving aqua regia comes from World War II and involves the Nobel Prize medals of two German physicists — Max von Laue (Nobel Prize in Physics, 1914) and James Franck (Nobel Prize in Physics, 1925).
After the Nazis rose to power in Germany, it became illegal to send gold out of the country. Fearing confiscation of their medals by the Nazi regime, von Laue and Franck sent their 23-karat gold Nobel Prize medals to the Niels Bohr Institute of Theoretical Physics in Copenhagen, Denmark, for safekeeping.
When Germany invaded Denmark on April 9, 1940, the medals were suddenly in danger. Hungarian chemist George de Hevesy, who was working at the institute at the time, came up with an ingenious solution — he would dissolve the medals in aqua regia. As de Hevesy later recalled: “While the invading forces marched in the streets of Copenhagen, I was busy dissolving Laue’s and also Franck’s medals.”
The 23-karat gold medals slowly dissolved into an orange-colored solution. De Hevesy placed the resulting flask on a shelf among many other similar-looking bottles and jars in the laboratory. When Nazi soldiers ransacked the institute, they searched for loot and evidence of wrongdoing, but paid no attention to what appeared to be just another jar of chemicals.
After the war ended, de Hevesy returned to Copenhagen and found the flask exactly where he had left it, completely undisturbed. He precipitated the gold out of the solution and sent it back to the Nobel Foundation in Stockholm. The foundation recast the medals from the original gold and presented them back to von Laue and Franck in 1952.
Remarkably, de Hevesy himself was awarded the Nobel Prize in Chemistry in 1943 for his pioneering work on radioactive tracers — making him a Nobel laureate who had quite literally held other Nobel Prizes in his hands.